
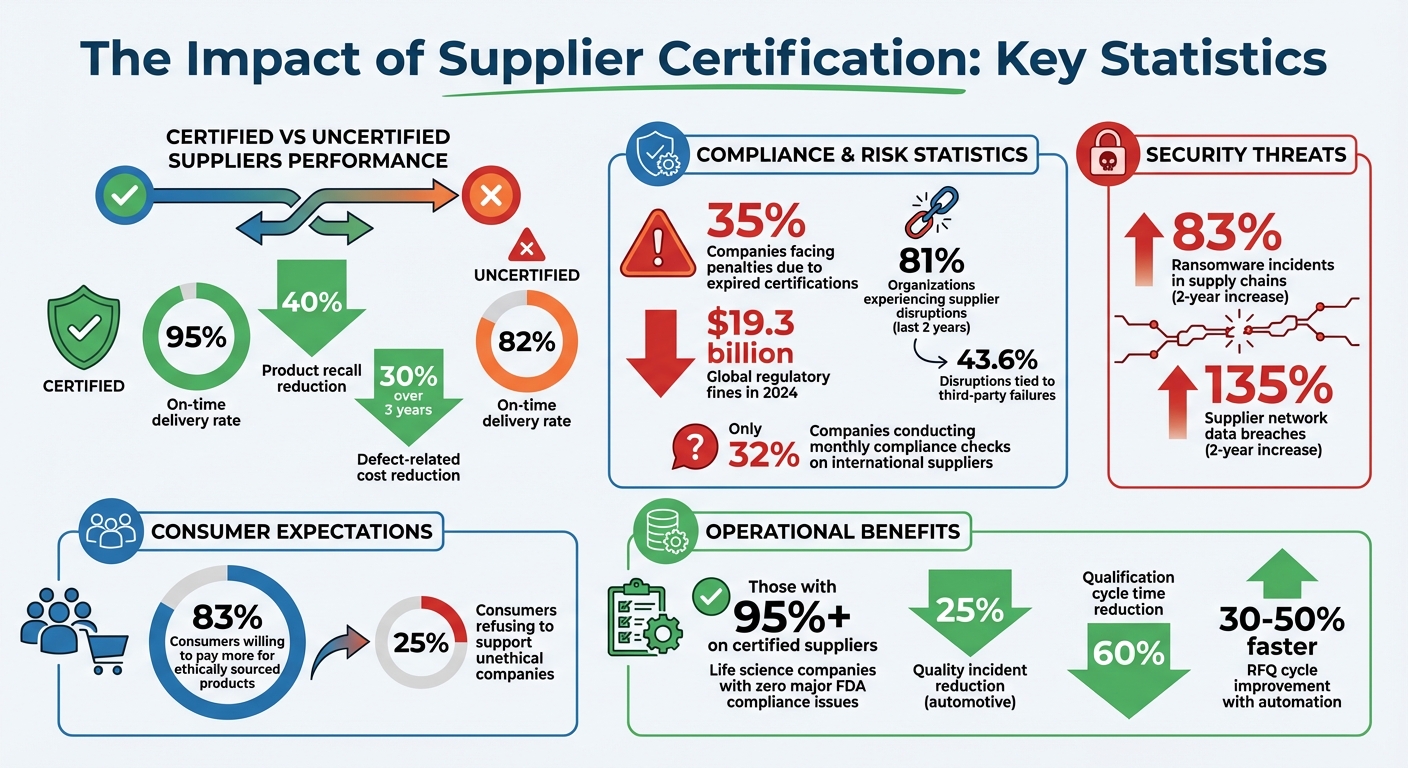
Supplier certification ensures vendors meet strict quality, regulatory, and operational standards. This process minimizes risks like compliance violations, supply chain disruptions, and reputational damage. Certified suppliers consistently outperform uncertified ones, with 95% on-time delivery rates and a 40% reduction in product recalls. In industries like aerospace, medical devices, and automotive, certifications such as AS9100, ISO 13485, and IATF 16949 are mandatory to meet safety and regulatory demands.
Key benefits of supplier certification include:
- Improved Product Quality: Certification ensures consistency and reduces defects.
- Legal and Financial Protection: Prevents regulatory fines and supply chain interruptions.
- Market Access: Enables entry into regions requiring certifications like CE or CCC.
- Operational Efficiency: Certified suppliers reduce disruptions and improve delivery reliability.
Relying on uncertified suppliers exposes businesses to risks like regulatory penalties, delayed production, and reputational harm. Modern tools like cloud-based platforms help automate certification management, ensuring compliance and reducing administrative burdens. Certification is not just a requirement – it’s a way to safeguard your supply chain and strengthen business performance.
Supplier Certification Impact: Key Statistics on Quality, Compliance, and Risk Reduction
Supplier Evaluation & Assessment How to Meet FDA QSR & ISO 13485 Requirements
sbb-itb-827f251
Risks of Using Uncertified Suppliers
Relying on uncertified suppliers can lead to compliance issues, production setbacks, and damage to your company’s reputation.
Regulatory Violations and Financial Penalties
When it comes to supplier actions, regulators hold companies directly responsible. Saying, "It happened at the supplier", won’t shield you from legal consequences. Take 3M, for example: in 2020, they were fined nearly $10 million after their suppliers conducted business with a sanctioned Iranian company. Beyond the financial hit, 3M faced significant operational challenges, including the need to replace suppliers on short notice and revamp their compliance processes.
Another case involved cosmetics company e.l.f., which paid a $1 million fine to the Office of Foreign Assets Control (OFAC) in 2019. The fine stemmed from importing eyelash kits that contained materials sourced from a sanctioned North Korean manufacturer. This violation was uncovered during an internal audit, underscoring the risks of working with suppliers that lack verified multi-tier transparency. Alarmingly, nearly 35% of companies have faced regulatory penalties because of expired supplier certifications, and global regulatory fines reached $19.3 billion in 2024.
But the risks don’t stop at fines. These compliance failures can also disrupt supply chains, making uncertified suppliers a liability.
Supply Chain Interruptions
Uncertified suppliers can destabilize operations in ways that go beyond financial penalties. Without proper change control systems, these suppliers may relocate facilities, alter materials, or adjust processes without warning – directly affecting product quality and disrupting validated workflows. This reactive environment means quality issues often surface only after production is delayed, customers complain, or recalls are necessary.
The numbers tell the story. Certified suppliers consistently achieve on-time-in-full delivery rates of 95%, while uncertified ones lag at just 82%. Poor handling of Corrective and Preventive Actions (CAPAs) by uncertified suppliers often leads to recurring issues and slow resolutions. Worse, if non-compliance is discovered – whether related to sanctions or labor practices – companies are forced to sever ties immediately and scramble for replacements to meet production demands. Shockingly, only 32% of manufacturing companies conduct monthly compliance checks on their international suppliers.
Damage to Company Reputation
The ripple effect of operational disruptions extends to your company’s reputation, especially as consumers increasingly demand ethical sourcing. A striking 83% of consumers say they’re willing to pay more for ethically sourced products, while 25% refuse to support companies involved in unethical practices. Partnering with suppliers that lack social compliance certifications, such as BSCI or SA8000, opens the door to allegations of unethical behavior, tarnishing your brand image.
Matthew Debbage, CEO of Creditsafe US and APAC, puts it bluntly:
"Many companies are simply running compliance checks to tick a box and show that they did the necessary due diligence. But they’re not using the results to protect the integrity of their global supply chains."
Additionally, uncertified suppliers can block access to key markets. Without region-specific certifications like CE for Europe or CCC for China, products may be barred from entering critical markets, limiting revenue potential and weakening your competitive edge.
Benefits of Supplier Certification Programs
Supplier certification programs go beyond simple compliance – they actively improve supply chain performance by addressing quality, legal, and market challenges. These programs create a structured approach to ensure suppliers meet rigorous standards, delivering measurable benefits.
Maintaining Product Quality and Consistency
Supplier certification plays a critical role in maintaining consistent product quality. By requiring raw materials and ingredients to meet strict standards, these programs ensure that production starts with a solid foundation. Independent audits verify a supplier’s ability to consistently meet these expectations.
For example, in May 2025, Westrock Coffee adopted SafetyChain‘s supplier compliance system to monitor the quality of pre-ground coffee beans. By analyzing grind size and yield data in real time, the company quickly resolved quality issues, boosting production time by 10% and improving quality control precision.
Certified suppliers also help reduce costly disruptions. Companies that implement strong certification programs have seen product recall risks drop by over 40%, while defect-related costs decreased by around 30% over three years. Certification frameworks like ISO 9001 ensure uniform production processes, preventing defects from reaching consumers and minimizing the risk of recalls.
Lowering Legal and Financial Risks
Certification programs also help protect companies from financial and legal risks by ensuring supplier compliance with regulations. Large businesses typically allocate 70–75% of their operating expenses to external suppliers, making supplier reliability crucial.
The financial stakes are growing. Over the past two years, ransomware incidents in supply chains have risen by 83%, and supplier network data breaches have surged by 135%. Regular financial evaluations of suppliers can identify potential insolvency risks, helping businesses avoid disruptions. In fact, 81% of organizations have experienced supplier-related disruptions in the last two years, with 43.6% of interruptions tied directly to third-party failures.
Certification programs often include contractual safeguards, such as "right-to-audit" clauses, minimum insurance requirements, and liability terms, which help prevent issues like payment fraud or unauthorized banking changes. With updated NACHA rules for 2026 requiring automated verification of account ownership for ACH payments, maintaining accurate documentation with certified suppliers ensures compliance.
Beyond reducing risks, certification enhances market opportunities and competitiveness.
Improving Market Access and Competitiveness
Certification can unlock access to markets that require specific approvals. For instance, CE marking is mandatory for selling in the European Economic Area, CCC certification is required for China, and FCC approval is essential for telecommunications products in the United States.
Beyond regulatory access, certifications provide a competitive edge. Quality certifications rank among the top six factors B2B buyers consider when selecting suppliers. With 70% of buyer research happening anonymously online, lacking recognized certifications like ISO 9001, IATF 16949, or AS9100 can lead to early disqualification.
Operational advantages are equally striking. Life science companies report zero major compliance issues during FDA inspections when over 95% of their procurement spend is directed to certified suppliers. Similarly, automotive manufacturers have seen a 25% reduction in quality incidents and a 60% decrease in qualification cycle times by using digital supplier management systems.
Judy Chen, Head of Marketing at SourceReady, emphasizes the importance of certifications:
"Valid certifications confirm that a supplier adheres to globally recognized industry standards… These frameworks promote consistent quality, ensure compliance with ethical practices, and reduce supply chain risk."
How to Implement Supplier Certification Processes
Creating an effective supplier certification program takes a well-organized approach that ensures both thoroughness and efficiency. The process revolves around three main activities: evaluating suppliers before entering into agreements, performing regular audits to confirm ongoing compliance, and maintaining meticulous documentation to avoid disruptions in your supply chain.
Evaluating and Selecting Suppliers
Before engaging with suppliers, it’s crucial to clearly define your product specifications and any regulatory requirements. Identify the certification standards relevant to your industry – such as ISO 9001 for quality management systems, RoHS compliance for electronics, or CE marking for goods sold in European markets. Use B2B platforms and import data tools to narrow down potential suppliers, and verify their legitimacy by checking business licenses and tax registrations through official government databases.
Take extra care when authenticating certifications. Always request original, high-quality PDF files and cross-check them against official registries like IAF CertSearch, the FDA VQIP directory, or the BRCGS Directory. Be on the lookout for signs of forgery, such as low-quality logos, missing expiration dates, or vague descriptions of what the certification covers.
"A factory that asks smart follow-up questions about your product is usually more capable than one that immediately quotes a price without understanding the details." – Jim Kennemer, Founder, Cosmo Sourcing
To ensure fairness and consistency in supplier evaluations, use a weighted scoring model. Assign points to various criteria: quality (35 points), technical fit (25 points), cost (15 points), environmental and social governance (10 points), delivery performance (10 points), and digital readiness (5 points). This approach reduces subjective bias and helps standardize decision-making across your procurement team. Companies using digital supplier management systems have reported a 60% reduction in qualification cycle times and a 25% decrease in quality issues.
This initial evaluation forms the groundwork for continuous audits and monitoring.
Performing Regular Audits and Assessments
After suppliers are onboarded, it’s essential to verify their compliance regularly. Certification isn’t a one-and-done process – it requires ongoing oversight. Schedule audits based on the risk level of each supplier. For instance, suppliers handling safety-critical components should undergo annual on-site factory audits, while medium-risk suppliers might need reviews every 24 months. Low-risk vendors can often be managed through annual desk reviews of their documentation.
When conducting factory audits, focus on quality control, equipment maintenance, and adherence to safety protocols. A general factory capability audit in Vietnam typically costs between $300 and $400, while more detailed social compliance audits can cost up to $1,000. Announced audits provide a snapshot of a supplier’s ideal operations, but unannounced visits can reveal their everyday practices.
Use scorecards to track supplier performance metrics like defect rates, on-time delivery, and responsiveness to quality concerns. Qualified suppliers often achieve on-time, in-full delivery rates of at least 95%, compared to just 82% for unqualified ones. Revalidate certifications annually or during contract renewals, and conduct full re-qualifications every three years or whenever significant changes occur in production processes or facility locations.
Managing Certification Documentation
Proper documentation management is the backbone of a successful supplier certification process. Keep all verified supplier documents in a centralized repository – ideally within an electronic quality management system that integrates with your ERP platform. This ensures that procurement, quality, and compliance teams can easily access the same up-to-date information. Verifying a single supplier’s documentation typically takes 8 to 20 minutes when following a standardized checklist.
Set up automated alerts to notify you of upcoming certification expirations, ideally 60–90 days in advance. This helps prevent sourcing from suppliers with expired certifications. Double-check that the legal name and address on each certificate match exactly with the details on purchase orders and contracts. Include documentation requirements in quality agreements, and ask suppliers to notify you of any material or process changes at least 30 days in advance.
Maintain an Approved Supplier List (ASL) that integrates directly with your purchasing system. This ensures that only vetted and qualified vendors receive purchase orders. Regulatory agencies like the FDA (21 CFR 820.50) and EU-GMP (Chapter 7) require version-controlled ASLs to track supplier performance and compliance history. Companies that combine supplier qualification with continuous monitoring often see defect-related costs drop by around 30% over three years.
Using Technology for Certification Management
Trying to manage certifications through spreadsheets and emails can leave room for compliance issues. Cloud-based platforms tackle this by automating every step of the certification process – from evaluating suppliers to ongoing monitoring and renewals. QSTRAT, for instance, transforms certification management into a seamless, proactive system that integrates directly with existing business workflows.
Automating Supplier Evaluation and Audits
Modern sourcing platforms simplify supplier evaluations with eForms, allowing suppliers to respond to certification requests without the hassle of manual data entry. QSTRAT takes this further by issuing RFx requests as electronic forms, which suppliers can complete using familiar tools. This reduces errors and ensures suppliers provide all necessary information consistently. Automated alerts also notify both procurement teams and suppliers when certifications are nearing expiration, streamlining renewal processes.
For example, Carl Zeiss uses QSTRAT to gather and analyze over 50 data points from suppliers during bidding and onboarding. What once took weeks of manual effort is now completed much faster. In fact, companies using automated workflows like this report RFQ cycles that are 30–50% faster than traditional methods.
"For BAE, QSTRAT Sourcing is much more than a great tool to compare price, labor, raw material, packaging, and transportation among our suppliers. It’s helping BAE become a better manufacturer by using QSTRAT Sourcing as a learning tool for suppliers. Through this process we are driving our quality initiatives forward." – BAE
The platform also ensures compliance with regulations by maintaining a SOX-compliant audit trail, complete with time, date, and user stamps for all changes and communications. This feature is invaluable during regulatory inspections. With over $5 billion in RFQs issued across 22 countries, QSTRAT has proven its ability to manage supplier certifications on a global scale. Additionally, its document management capabilities keep all certification data easily accessible.
Centralizing Certification Records
Automation is only part of the equation – centralizing certification records takes compliance management to the next level. A centralized repository acts as a single source of truth, housing all supplier communications, certifications, and audit records in one secure location. This means that during audits, your quality team can quickly access the latest certification without wasting time digging through scattered files. For instance, Cloetta, a confectionery manufacturer, achieved 100% supply chain compliance by adopting a digital platform to centralize supplier certifications.
Cloud-based systems also shift compliance management into a proactive mode. These platforms can flag issues in real time, such as when a supplier is added to an international blacklist or when a certification is revoked. This allows businesses to address problems before they disrupt production. Automated tracking of expiration dates ensures that certifications, insurance policies, and NDAs are renewed on time, avoiding compliance gaps that could lead to penalties.
"Supplier compliance software… ensures every vendor meets your standards and obligations, so you’re not always worrying about surprise audits or issues." – Richard Teuchler, Head of Demand Generation, Kodiak Hub
Connecting Certification Processes with ERP and CRM Systems
Integrating certification processes with ERP and CRM systems creates a seamless flow of data, building on the benefits of automation. QSTRAT’s architecture supports integration with platforms like MFG/PRO, enabling certification status to control supplier eligibility for purchase orders. For instance, if a supplier’s ISO certification expires, the system can automatically block new purchase orders until updated documentation is provided.
This integration eliminates duplicate data entry and ensures consistency. Once a supplier completes onboarding and certification in QSTRAT, the validated information automatically populates the ERP’s approved supplier list, no manual input required. One QSTRAT client shared their experience: "By leveraging QSTRAT’s suite of products with our existing MFG/PRO and Cyberquery solutions we can now accurately quote, track successes and measure our accuracy of our projected costs versus our true costs".
Conclusion
Supplier certification plays a crucial role in managing risks, protecting your company from regulatory breaches, supply chain disruptions, and damage to your reputation. With over 70% of Chief Procurement Officers highlighting a rise in procurement-related risks and supply chain interruptions for 2024–2025, the pressure to act has never been greater. Certifications like ISO 9001, CE, and RoHS serve as clear evidence that your suppliers meet essential quality, safety, and environmental standards. This helps reduce defects and ensures consistent product quality throughout your operations. To address these challenges effectively, businesses need a modern, integrated approach to managing certifications.
"Regulators no longer accept the argument that ‘the issue occurred at the supplier.’ The legal and compliance responsibility remains with the purchasing organization." – Wali Alam, Founder, Quality Institute of America (QIA)
As discussed earlier, adopting a proactive compliance system is critical. Leveraging modern technology takes this protection even further. Moving away from spreadsheets to technology-driven certification management turns compliance into a strategic advantage. Companies that adopt such systems report 30–50% faster RFQ cycles while maintaining complete audit trails.
QSTRAT’s cloud-based platform simplifies the certification process by automating evaluations, centralizing records, and linking certification status directly to purchase order eligibility. This provides real-time visibility into supplier compliance, reducing administrative burdens and identifying high-risk suppliers before they cause disruptions.
By embracing this tech-forward approach, companies not only cut down on administrative work but also strengthen their supply chain against potential risks.
"Compliance is Not a Cost – It’s an Advantage" – Judy Chen, Head of Marketing, SourceReady
The businesses that succeed in today’s stringent regulatory environment are those that view supplier certification as an ongoing strategic effort, not a one-time task. Using technology to enable continuous monitoring, verify credentials at their source, and conduct risk-based audits turns certification management into a tool for gaining a competitive edge. This approach opens up opportunities in new markets and builds lasting customer trust.
FAQs
What supplier certifications do I actually need for my industry?
Certifications vary depending on your industry and the regulations you need to follow. For instance, the aerospace sector often requires AS9100 Rev D or AS9120B to meet safety and quality standards. In other industries, certifications like ISO 9001 (focused on quality management), ISO 14001 (related to environmental management), or FDA compliance might be essential.
It’s important to align your certifications with regulatory requirements, customer expectations, and your overall approach to risk management. Tools such as QSTRAT can simplify the certification process, helping you stay compliant while saving time and effort.
How often should suppliers be re-audited or re-certified?
Suppliers need to undergo regular re-audits or re-certifications. How often this happens depends on factors like industry standards, the level of risk involved, and specific certification requirements. For most cases, annual audits are a good rule of thumb. However, industries with higher risks or stricter regulations might need more frequent evaluations to maintain compliance and address potential risks properly.
How can I avoid purchasing from suppliers with expired certifications?
To steer clear of working with suppliers whose certifications have expired, it’s crucial to stay on top of their records. Keep a current database of certifications, schedule regular reviews, and conduct supplier audits to ensure everything is in order. Leveraging supplier management tools can make it easier to track certification statuses and stay compliant. By actively keeping an eye on expiration dates, you can minimize the risk of engaging with suppliers who don’t have valid credentials.



